Why Turnkey Project in Pharmaceutical Industries
Pharmaceutical turnkey projects provide a complete solution for drug manufacturers, encompassing everything from initial design to final production, resulting in faster time-to-market and reduced operational costs.
End-to-end solutions in pharmaceutical turnkey projects can result in cost savings of up to 30% compared to traditional project delivery methods, while also reducing time-to-market by up to 40%.
Generally speaking, the higher the turnkey degree of a project, the more cost savings will be achieved, and the less ineffective communication and mismatches between different suppliers’ products will occur. The design team of Senieer will fully cooperate with your project and provide the most suitable solution based on the actual situation.
Key Stages of Pharmaceutical Turnkey Projects
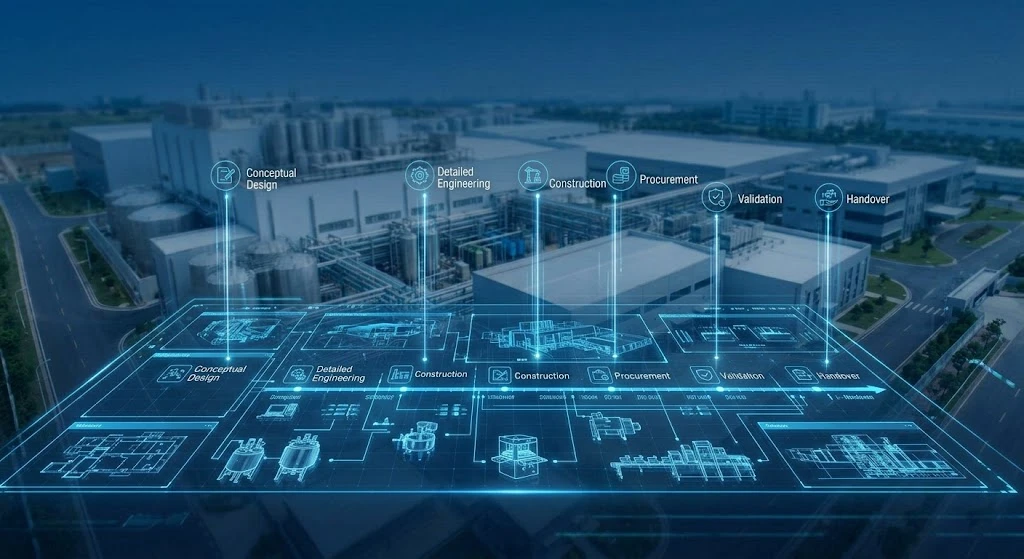
Chapter 1:Conceptualization and Feasibility Studies
During this phase, the contractor works closely with the client to define project goals, assess technical and financial feasibility, and develop a preliminary design concept. This stage sets the foundation for the entire project and helps identify potential challenges early on.
Chapter 2:Detailed Design and Engineering
architects and engineers create comprehensive blueprints for the facility, including specifications for equipment, utilities, and process flows. This stage also involves obtaining necessary regulatory approvals and ensuring compliance with GMP standards.
A well-executed design phase in pharmaceutical turnkey projects can reduce construction time by up to 25% and minimize costly changes during later stages of the project.
Chapter 3:Construction and Preparing of Facility
Once the design is finalized, construction begins. This stage involves site preparation, building construction, and the installation of critical systems such as HVAC, electrical, and plumbing. Throughout the construction phase, project managers closely monitor progress to ensure adherence to timelines and quality standards.
Chapter 4:Procurement
Sourcing and supplying processing machinery, packaging lines, and production equipment according to the objectives of the project within the defined timeline.
Chapter 5:Equipment Installation
In this stage, the project goes through the FAT. The FAT of the processing machines includes the approval from the regulatory authority for the installed machines, and the project gets the pre – approval for the production line.
Chapter 6:Commissioning and Qualification
In this stage of a turnkey project, it involves commissioning, qualification, and validation. During commissioning, all systems and equipment are tested to ensure they function as intended. Qualification involves documenting that the facility, equipment, and processes meet predefined specifications.
Finally, validation demonstrates that the facility consistently produces products that meet quality standards.
Chapter 7:Validation and Regulatory Approval
Ensuring the plant meets regulatory standards (cGMP, USFDA, WHO-GMP) through documentation, testing, and commissioning.
Effective regulatory compliance strategies in pharmaceutical turnkey projects can reduce the time required for regulatory approvals by up to 50% and minimize the risk of costly remediation efforts.
Why choose Senieer
Senieer are known all over the world for our pharmaceutical equipment. We have over 37 years of pharmaceutical experience and have assisted 61 countries in solving pharmaceutical issues.
Senieer provide complete design, engineering, construction, start-up of new pharmaceutical facilities, even assisting the Clients through scouting activities.
Senieer project drivers satisfy all pharmaceutical regulatory and QA requirements, aligning the final product to the international Pharmacopoeias and capitalizing upon a careful, customer-oriented project management
Senieer offer a comprehensive End-to-End Solutions package that addresses all aspects of facility design, construction, and commissioning.
Key Stages of Pharmaceutical Turnkey Projects
- Chapter 1:Training & Handover
- Chapter 2: Technical Expertise and Knowledge
- Chapter 3: Industry Experience and Track Record
- Chapter 4: Global Reach and Supply Chain Capabilities
- Chapter 5:Financial Stability and Resources
- Chapter 6:Digital Tools and Project Transparency
- Chapter 7: Tools used By Seineer

Chapter 1:Training & Handover
Providing staff training and full documentation before handing over the operational facility.
Senieer provides comprehensive equipment documentation, Including training, operation, and maintenance manuals, in compliance with the strictest standards of the pharmaceutical industry. We offer a complete set of cGMP documents including Qualifications(DQ,IQ, OQ,PQ ) , FAT, SAT, USER manual, etc. We offer certifications such as CE, GMP,cGMP, ISO9001, FDA, WHO, etc., to comply with global quality, safety, and regulatory requirements.
Choosing the right partner for a pharmaceutical turnkey project can lead to cost savings of up to 20% and reduce project timelines by up to 30% compared to working with less experienced contractors.
Chapter 2:Technical Expertise and Knowledge
- Expert Staff of Engineers in different fields including Civil , Electrical and mechanical engineers
- Regulatory Compliance Expertise with different Awareness of regulation requirements from different authorities like FDA , EMA and WHO
- Executive Project Managers and executive team with deep understanding of pharmaceutical manufacturing processes, GMP requirements, and the latest technologies relevant to your project.
Chapter 3:Industry Experience and Track Record
- Senieer has more than 37years’ experience in manufacturing and delivering different machine and equipment in pharmaceutical and food industries
- More than 500 machine had been installed and validated over 61sites in different countries (Asia , Europe and Africa )
- Hongkong Project and Malaysia Projects
- Expert Customer service team working 24/7 to receive all customers complaints and inquires
Chapter 4:Global Reach and Supply Chain Capabilities
- Large Data of several certified suppliers of all the required machines, Equipment, Utilities, systems, raw materials with proven track of high-quality product.
- Proven track record in the pharmaceutical industry and proven history of successfully completing several projects with different scale and complexity in different country .
Chapter 5:Financial Stability and Resources
Senieer business sections are operating under the strict International Quality Standard of operation with control regulation certificate ISO9001 which is called the “International Quality Certifications”.
If you’re in need of any help, and want to bring your factory’s pharmaceutical productivity to a new level with 4.0 industry solution from pharmaceutical company, letting Senieer booms and supports your business right now.
Senieer always thinks of the dignity of customer and promises stabilized services. We have a passion for success of customers together.
Chapter 6:Digital Tools and Project Transparency
- Real-time project tracking,
- BIM(Building Information Modeling )based design
Future-focused pharmaceutical turnkey projects that incorporate flexible design and sustainable practices can reduce operational costs by up to 25% over the facility’s lifetime and improve overall equipment effectiveness by up to 15%. Integrated ERP/CRM systems to keep you informed about the project 24/7.
Chapter 7:Tools used By Seineer
- IIoT Gateways and Sensors:Applying Devices that equipped with sensors for monitoring parameters like temperature, vibration, and energy, with data often processed on-site via edge computing to minimize latency.
- Cloud Computing and Data Platforms:applying system for storage of Real-time data with analyzing in the cloud, allowing for remote monitoring, and advanced analytics, providing insights into production processes.
- Robotics and Automation:Sourcing of required robotics and Automation in different area of Warehouses and Production that reduces manual labor and improves precision.
- AI for Maintenance and Quality: Sourcing AI system for Machine learning algorithms which improve predictive maintenance (PdM) to prevent downtime and for automated visual inspection to improve quality control.
- 3D Modeling & CAD Software:Used for detailed engineering, facility layout, and 3D visualization.
- Computational Fluid Dynamics (CFD):For modeling HVAC, cleanroom dynamics, and energy consumption.
- Digital Twins:Used to simulate process flows and utility systems before construction.










